2023 and the start of 2024 in research were full of illuminating studies and earthquaking...
Why drug development is slowing down – and what to do about it

When the FDA issued emergency authorization use for the first COVID-19 vaccine in December 2020, it marked just 10 months since the Secretary of the Department of Health and Human Services officially declared a public health emergency.
Scientists estimate that in this instance, compared to the traditional vaccine creation process, approximately four years got chopped off the development timeline.
But the COVID-19 vaccine was an exception—an extraordinary circumstance. The urgency of the pandemic directed unprecedented attention and resources towards development of the vaccine. 43,000 volunteer testers signed up over the course of a mere 16 weeks. And the typical 10-month FDA review period was reduced to just three weeks.
Despite this remarkably fast turnaround for the COVID vaccine, by and large, drug development has slowed the last few decades. Since 2017, only 12 antibiotics have been approved worldwide, 10 of which belong to existing bacterial classes with established antibiotic resistance. On average, resistance is developed to new market entrants just 2-3 years post-market entry.
Nowadays, the average time-to-market for a new drug is 12 years. Failure rates are as high as 90%. And average R&D costs of $1.1 billion twenty years ago have soared to $4.4 billion.
Developing new lifesaving, innovative pharmaceuticals is more difficult than ever, and no one factor is to blame. In this article, we explore the diverse reasons for this lag, ongoing solutions in the field, and the role of proactive research.

Is all the low-hanging fruit picked?
Another unfortunate trend in drug development is that many new drugs simply are not effective. In recent years, the percent of medicines that needed to be pulled from the market has increased as much as 25%. In 2022, the FDA approved 37 novel drugs, the lowest number since 2016.
Some researchers argue that science has currently maxed out medicines that can be made based on our current understanding of the body. In other words, the “low-hanging fruit has already been picked,” a claim heard throughout the biopharmaceutical research community. The claim is mostly based on observing the historical trends of straightforward and obvious drug targets being addressed, and the diminishing returns of the drug development world.
However, there is little research out there rigorously defining what it would mean for all the low-hanging fruit to be picked, and whether or not that is the case. Other scientists point to evidence suggesting that there are plenty of reasons that drug development should not need to slow down in the current scientific climate. The main reasons include:
- Basic research on biology, genetics, and disease mechanisms is continuing to advance, especially through technologies such as CRISPR gene editing and Single-Cell RNA sequencing
- There are many rare and neglected diseases that still require targeted drug development, such as ALS, Huntington’s Disease, Chagas Disease, and African Sleeping Sickness
- Advances in approaches to drug-development, such as the rise of personalized medicine and international collaboration, give rise to new possibilities and approaches
Legal and administrative issues
Commentators often point to regulatory bodies as a source of woe slowing down pharmaceutical research. Regulatory bodies do set stringent requirements and complex processes, including multiple stages of testing.
On the other hand, the FDA approves drugs much faster than it did 30 years ago. However, studies have demonstrated that faster doesn’t mean better or more useful: a study in the British Medical Journal showed that expedited pathways created by the FDA did not lead to an increase in innovative drugs coming to market.
Even with overall approval speed on the rise, there are regulatory issues that have grown in complexity throughout the 21st century:
- Complexifying requirements and processes: Regulatory approval pathways require multiple stages, including preclinical testing, clinical trials, and regulatory submissions. Fast track, accelerated approval, and priority review programs have been increasingly utilized over the last 20 years, creating a more flexible but also complex approval process.
- Intellectual property lawsuits: Patent issues and IP disputes can hinder the development of drugs when multiple companies are working on similar therapies. For example, in recent years AbbVie had a patent dispute with Boehringer Ingelheim over biologics used in treating autoimmune diseases, such as rheumatoid arthritis. AbbVie has also used patent law to exclude biosimilar entries since then.

Pharmaceutical industry market incentives
If there’s little reason to believe drug development should be slowing down simply because the science is getting harder or that the approval takes more time, eyes will naturally turn to the the economic and market structure ruling pharma companies.
A study published in the British Medical Journal by Donald Light and Joel Lexchin argue that the business model for pharmaceutical companies does not focus on breakthrough research discoveries. They state that the enemy of innovation in the industry is the way companies emphasize marketing and defending existing blockbuster products. According to the study, companies spend only 1.3% of revenues on discovering new chemical entities, compared to 25% on promotion.
The article also disputes claims of how costly it is to develop new drugs. The study puts forth that half of the hefty billion-dollar estimates to discover a new chemical entity “comes from estimating how much profits would have been made if the money had been invested in an index fund of pharmaceutical companies that increased in value 11% a year, compounded over 15 years.”
Several studies have demonstrated that pharmaceutical companies tend to invest in therapeutic areas with the largest markets and profit margins, diverting attention from innovation.
Other critics have argued that current incentives in the pharmaceutical industry are skewed towards “me too” drugs, minor variations on existing treatments. Currently, the FDA is making the most progress approving generic and biosimilar drugs; which, while still signifying excellent developments, does not represent genuinely disruptive discoveries.
Lack of reproducibility
Reproducibility has become a major topic for the science community. Studies in recent years have shown that 60% of researchers could not reproduce their own findings. As evidence of this, only top-tier journals have statistical editors that assess whether p values in published studies are credible. (P values measure the probability of getting the same results by chance alone.)
Commonly cited sources of this scientific crisis include a lack of access to methodological details and raw data, use of cross-contaminated materials, poor research practices, the inability to manage complex datasets, and cognitive bias.
The striking lack of reproducibility in science means that researchers cannot always rely on published data from a different lab. But the use of outside studies to make innovative discoveries is core to both drug development and science as a whole.
One final factor potentially slowing down drug development besides reproducibility is the COVID-19 pandemic. Scientists have reported that COVID-19-related research often took the priority over their other topics of interest, and this trend also extended to the pharmaceutical world.
Solutions
A lot of scientific effort and collaboration has gone into identifying whether or not drug development is slowing down, and if so, how. Along with such efforts have come promising solutions to boost innovation and new discoveries.
-
Innovative trial designs
New and evolving trial designs have the promise to empower the pharmaceutical industry. A study conducted by Economist Intelligence Unit identified four designs proven to have a demonstrated impact on innovation (a 10-21% higher Phase I and II likelihood of launch compared to drugs developed using current practices):
- Adaptive trials: Allow certain planned modifications to be made to the design study after the trial’s initiation. For example, a trial could allow certain treatments to be added or dropped depending on the interim results.
- Patient-centric trials: Make participation as convenient as possible for patients and ensuring patients’ needs are met, i.e. using remote technology to reduce clinic visits. It also ensures that trial outcomes are meaningful and relevant to patients.
- Precision medicine trials: Evaluate the effectiveness of treatments in specific subgroups of patients, narrowing down based on demographics, environment, and lifestyle. For example, a study could select specific biomarkers or molecular subtypes.
- Real-world data trials: Use evidence from real-world data (RWD), including electronic health records, medical claims, product registries, and data generated by patient’s health and electronic devices.

-
Translational and proactive research
Translational research takes basic scientific discoveries and converts them into results that are beneficial to human health and society. While reproducibility, as discussed above, is a problem, starting with established scientific results and then building on them for new drug discoveries has been hailed as a promising approach.
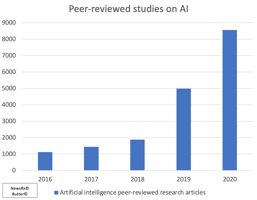
Meanwhile, proactive research uses artificial intelligence to automatically locate relevant studies, clinical trials, and materials and keeps researchers and clinicians on top of the latest scientific discoveries without wasting time searching for information. By making science less reactive—more focused on the big picture than immediate needs—and more results-driven—less time spent evaluating the quality of studies or mining journals for content—this approach accelerates the research process.
-
Leveraging data and technology
AI can also be leveraged not only to conduct basic and proactive research, but at the heart of the drug development process. So far developers have had success utilizing automation, AI, and machine learning in trial design, drug target identification and validation, predictive modeling, and statistical analyses.
Previously, uses of automation and large data were limited, as data was kept locked up in different functional groups and organizations. But with better cloud-based data management systems, organizations can securely exchange data—opening the door to leveraging big data and automated insights.
According to Boston Consulting Group, as of March 2022, biotech companies using an AI-first approach saw a growth rate in the number of molecule drugs in the clinical trial stage by nearly 40% year-over-year.
-
Fixing market incentives
Light and Lexchin argued for a few regulatory and market solutions to address lack of innovation. First, they recommended that regulatory bodies should stiffen the requirement for new drug approvals to incentivize genuine innovation as opposed to minor upgrades. Second, they supported a policy that would give large taxpayer-funded cash prizes to companies that deliver real improvements in healthcare, but allow generic competition to begin immediately (instead of companies being allowed to charge high prices for new products).
Such an approach would be disruptive and controversial. On a smaller scale, governments and philanthropic organizations have seen some success in sparking innovation through new funding initiatives, including the lump-sum payment method as discussed by Light and Lexchin above. Another idea is a subscription-style payment model, where the government pays pharmaceutical companies upfront for access to drugs based on their usefulness.
Overall, fixing market incentives will require pharmaceutical companies’ success to be tied to health outcomes rather than profits. Tying R&D and executive compensation to health outcomes is one suggestion, but choosing metrics to define these outcomes is challenging. Others have proposed tying sales representative compensation to patient outcomes.
Sparking pharmaceutical innovation is a heavily debated topic, and a truly productive future will surely require some combination of all these initiatives. In just the last decade, there have been over 50 major international and national initiatives aimed to incentivize antibiotic innovation—and there’s plenty of political momentum and willpower to do so.
In recent years, the antibiotic pipeline has been reinvigorated thanks to these efforts. But despite a larger number of antibiotics in the clinical pipeline, approvals are still low, with just three antibacterial drug launches since 2020.
It will be a combination of technological innovations and economic incentive-based solutions that together revitalize the areas of drug discovery that have slackened. A new era of drug development and human health is possible—unleashed by the potential of artificial intelligence and the willpower of human beings.

.jpg?width=50&name=DSC_0028%20(1).jpg)